Over 4.5 billion years ago, the Earth formed, initially enveloped in an atmosphere rich in hydrogen, helium, and water vapor. As the planet cooled, the water vapor condensed to form oceans, leaving behind an atmosphere composed of gases including CO2, nitrogen, and methane. Around 4.4 billion years ago, the Earth's molten surface solidified to form the first crust, marking the end of the planet's early molten state.
During the initial million years post its formation, the Earth's atmosphere might have contained CO2 concentrations as high as 10,000 ppm, primarily because there were no life forms to absorb CO2 through photosynthesis. Around 4.3 to 4.4 billion years ago, the Earth had cooled sufficiently for water to condense and form oceans, as evidenced by zircon crystals from that period.
Life began to emerge between 3.5 and 3.7 billion years ago, with the earliest direct evidence being fossilized bacteria from this time. This suggests that conditions conducive to life had existed for hundreds of millions of years before the first life forms appeared. The early atmosphere was dominated by gases such as water vapor, nitrogen, carbon dioxide, methane, ammonia, and hydrogen, with a notable absence of oxygen.
In the first few hundred million years, CO2 levels were exceedingly high, possibly over 5000 ppm, due to extensive volcanic outgassing and minimal absorption by rocks and emerging life forms. Around 2.7 billion years ago, the advent of cyanobacteria, capable of photosynthesis, began to gradually reduce CO2 levels. By the end of the Archean eon, approximately 2.5 billion years ago, CO2 levels had potentially decreased to about 4000 ppm, a hundred times the present levels. During the early Proterozoic era, the CO2 concentration remained between 10 and 100 times higher than today's levels.
As life continued to evolve, the decline in CO2 levels accelerated, with plants and algae playing a significant role in this reduction through the process of photosynthesis, where they consumed CO2 to produce oxygen and food, releasing oxygen back into the atmosphere. Between 600 and 400 million years ago, the Earth experienced another phase of extremely high CO2 concentrations, exceeding 6000 ppm. This period, characterized by a warmer climate, saw the flourishing of primitive plant life forms that thrived in the carbon-rich environment.
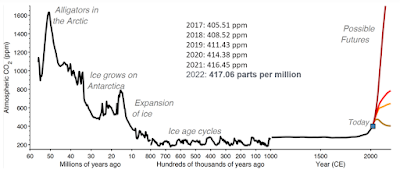
Around 50 million years ago, during the Eocene epoch, the Earth experienced a significant decline in atmospheric CO2 levels. This period is known for a series of drastic changes in the Earth's climate and environment. The movement of Earth's plates led to the uplift of mountain ranges, which increased weathering rates when CO2 was consumed from the air and converted into carbonate rocks. The spread of grasslands increased the rate of weathering of continental rocks. A reduction in volcanic activity and increased carbon sequestration in the deep sea, as well as the formation of the Antarctic ice sheet as well as spread of new types of phytoplankton could have also contributed.
The decline in CO2 levels during this period is associated with a general cooling trend, which eventually led to the ice ages of the more recent geological past. It's a complex interplay of geological, biological, and climatic factors that contributed to the dramatic fall in CO2 levels during this period.
The Holocene epoch, which began around 11,700 years ago and continues to the present day, is often characterized by relative climatic stability and prosperity, especially when compared to the fluctuating climates of the preceding Pleistocene epoch. The stable climate of the Holocene facilitated the rise of ancient civilizations, including Mesopotamia, Ancient Egypt, the Indus Valley Civilization, and others. These civilizations were able to develop complex societies, with advancements in technology, art, and architecture. The Holocene has also been a period of rich biodiversity, with a wide variety of flora and fauna flourishing in various ecosystems around the world.
While the debate continues regarding our entry into a potentially less stable epoch known as the Anthropocene, there is a concerted human effort to address the escalating levels of CO2 in the atmosphere through innovative technologies.
Direct Air Capture (DAC) stands as a pivotal technology in the array of Negative Emission Technologies (NETs) aimed at mitigating the escalating levels of CO2 in the atmosphere. This technology encompasses various systems including absorption and adsorption methods, which are currently the most mature and extensively researched approaches. These systems function by capturing CO2 directly from the atmosphere, either storing it to reduce long-term environmental impact or utilizing it in other chemical processes, thereby fostering a human-controlled carbon cycle. However, the nascent stage of this technology presents a spectrum of costs and energy consumption values, necessitating further research and development to enhance efficiency and economic viability. Key performance indicators (KPIs) such as thermal and electrical energy consumption, operational and capital expenditures, and environmental impact serve as critical metrics in evaluating and advancing DAC technologies. As the scientific community and industries strive to refine these technologies, the focus remains on optimizing various factors including the energy required for regeneration, the binding affinity of sorbents and solvents to CO2, and the design of air contactors.
In this evolving landscape, the Holocene company emerges as an important player, contributing towards achieving global climate goals.
Holocene is a startup based in Knoxville that is focused on developing and building plants capable of removing carbon dioxide from the atmosphere. The company has licensed a sustainable chemistry developed at the Department of Energy’s Oak Ridge National Laboratory (ORNL) for capturing carbon dioxide directly from the air. This technology utilizes a water-based, low-temperature process that employs an aqueous solution containing Bis-iminoguanidine (BIGs) receptors to absorb CO2, which then transforms into an insoluble crystalline salt that can be easily separated from the solution.
In the ever-evolving sphere of climate technology, accolades and recognitions serve as testament to the relentless efforts and innovations that companies bring to the fore. The Holocene company, despite not clinching the top spot at today's Innov865 annual pitch competition, managed to leave a lasting impression with their compelling and well-articulated pitch. Their technology, already a recipient of the prestigious R&D 100 Award, stands out in the quest to curb carbon emissions, promising a cleaner, greener tomorrow for generations to come.
REFERENCES
James W.B. Rae, Yi Ge Zhang, Xiaoqing Liu, Gavin L. Foster, Heather M. Stoll, Ross D.M. Atmospheric CO2 over the Past 66 Million Years from Marine Archives. Whiteford Annual Review of Earth and Planetary Sciences 2021 49:1, 609-641
Leonzio G, Fennell PS, Shah N. Analysis of technologies for carbon dioxide capture from the air. Applied Sciences. 2022 Aug 19;12(16):8321.
Kasturi A, Jang GG, Akin AD, Jackson A, Jun J, Stamberga D, Custelcean R, Sholl DS, Yiacoumi S, Tsouris C. An effective air–liquid contactor for CO2 direct air capture using aqueous solvents. Separation and Purification Technology. 2023 Nov 1;324:124398.
bigideaknox.com
https://knoxec.com/what-we-do/programs/wtbi/